Electron Configuration: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 6s2
Name |
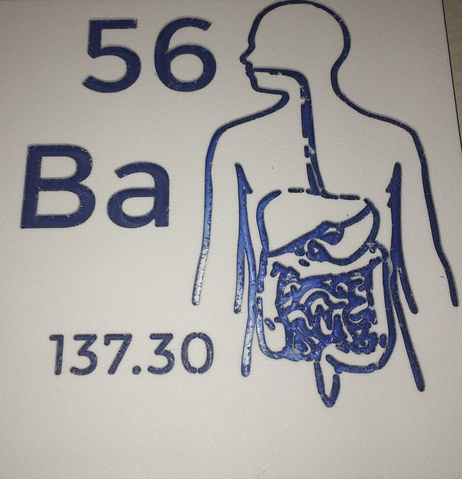
Barium |
Symbol |
Ba |
Atomic Number |
56 |
Atomic Mass |
137.30 |
Number of Protons |
56 |
Number of Electrons |
56 |
Number of Neutrons |
81 |
Melting Point |
727 degrees Celcius |
Boiling Point |
1897.0 degrees Celcius |
Normal Phase |
Solid |
Family |
Alkaline Earth Metal |
Period |
6 |
Group |
2 |
Origin of Name |
Barium comes from the Greek word, "barys", meaning heavy |
Date and Place of Discovery |
Isolated and identified in 1808 by Sir Humphrey Davy |
Interesting Facts |
|
Common Uses |
|